|
1/20/2024 0 Comments The ER has many complex proteins and so have a robust system for heat-shock protein assisted folding Here, we review recent advances in understanding the mechanism of protein translocation and transmembrane domain insertion in the ER, summarize new insights into selective cargo packaging, and discuss the roles of ER morphological dynamics in these processes.ĬOPII Cargo receptor Endoplasmic reticulum Membrane insertion Protein translocation Retrotranslocation.Ĭopyright © 2021 Elsevier Ltd. Some of these proteins are packaged into coat protein complex II-coated vesicles for export. They are then modified post-translationally and folded in the ER. These polypeptides enter cotranslationally in the ER lumen, which contains resident molecular chaperones and folding factors that assist their maturation. These proteins are translated by ribosomes outside the ER and require subsequent integration into or translocation across the lipid bilayer of the ER. The abundant, essential, cytosolic Hsp90 (Heat shock protein, 90 kDa) facilitates the folding and activation of hundreds of newly synthesized or misfolded. A substantial fraction of eukaryotic gene products are synthesized by ribosomes attached at the cytosolic face of the endoplasmic reticulum (ER) membrane. 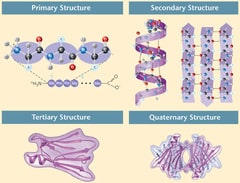
Through a continuous membrane network of sheets and tubules, the ER hosts secretory proteins, integral membrane proteins, and luminal proteins of the endomembrane system. The endoplasmic reticulum (ER) is the main harbor for newly synthesized proteins in eukaryotic cells. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
